P – Block Elements Class 12 Notes

Class 12 Chemistry Chapter 7 P – Block Elements Notes- Pdf Download
Chapter 7 P – Block Elements
Chapter 7 of the NCERT notes for Grade 12 Chemistry discusses general principles and procedures for element isolation that may be useful to students. This is one of the important chapters of grade 12 chemistry. These NCERT notes are designed according to the latest curriculum. So students can follow the latest NCERT solutions to help them prepare for the exam.
Elements from groups 13 to 18 of the periodic table are p-block elements. They have the valence shell electron configuration ns2np1–6. Since we discussed the elements of groups 13 and 14 in the previous class, we will therefore cover the remaining element groups of the p block in this chapter.
CBSE CLASS 12th CHEMISTRY 7 NOTES
Points to Remember
NCERT notes for Grade 12 Chemistry Chapter 6: Important Points to Remember
The entire scientific and technological process used for the extraction of pure metal from its ore is known as metallurgy.
All minerals are not ores, but all ores are minerals.
Some of the important procedures for the concentration of ores are Hydraulic washing, magnetic separation, froth floatation method and leaching.
In this process, the roasted or calcined ore is mixed with a suitable quantity of coke or charcoal (which acts as a reducing agent) and is heated to a high temperature above its melting point. During reduction, an additional reagent (flux) is also added to the ore to remove the impurities still present in the ore. The flux combines with the impurities to form a fusible product known as slag.
Certain metals, such as silver and gold, are extracted by dissolving the concentrated ore in some suitable reagent and then recovering the metal from the solution by treatment with some more electropositive metal. Such a process in which metal is recovered from an aqueous solution is referred to as hydrometallurgy.
Topic and Sub-Topics
Students will need to ensure that they prepare properly for the exams. NCERT notes for Grade 12 Chemistry Chapter 6 can be beneficial for students. Students who are struggling or having difficulty can refer to the videos on Vidyakul to prepare for their exams.
It is essential that students refer to the subtopics of the chapter to prepare for the exams. Students should refer to all questions for each subtopic. This will help students prepare for the exams more easily. In the table below, we have provided a full range of subtopics for your convenience.
Download this solution for FREE Download This PDF
Download Vidyakul App for more Important videos, PDF's and Free video lectures.












Important Links:
Few Important Questions
How many elements are present in the p-Block?
There are 35 p-block elements, all of which are in p orbital with valence electrons.
What is a ‘Halogen’?
Any of the five elements fluorine, chlorine, bromine, iodine and astatine that form part of group VII A of the periodic table and exist in the free state normally as diatomic molecules.
What is ‘Oxoacid’?
An oxoacid (oxyacid) is an acid that contains oxygen.
Practice Questions
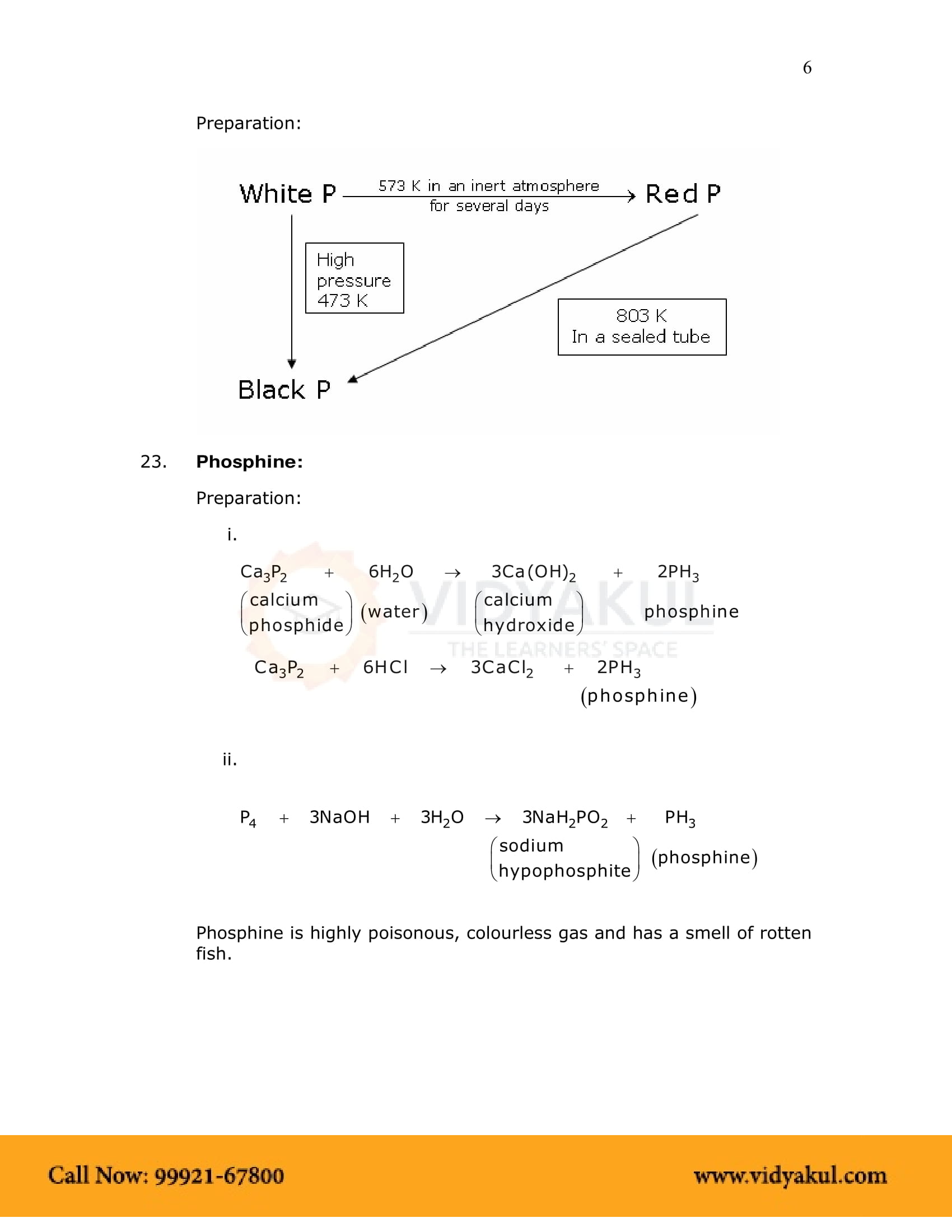
Distinguish between red phosphorus and white phosphorus.
How many oxoacids does fluorine form? Why?
Explain the manufacturing of sulphuric acid by the contact process.
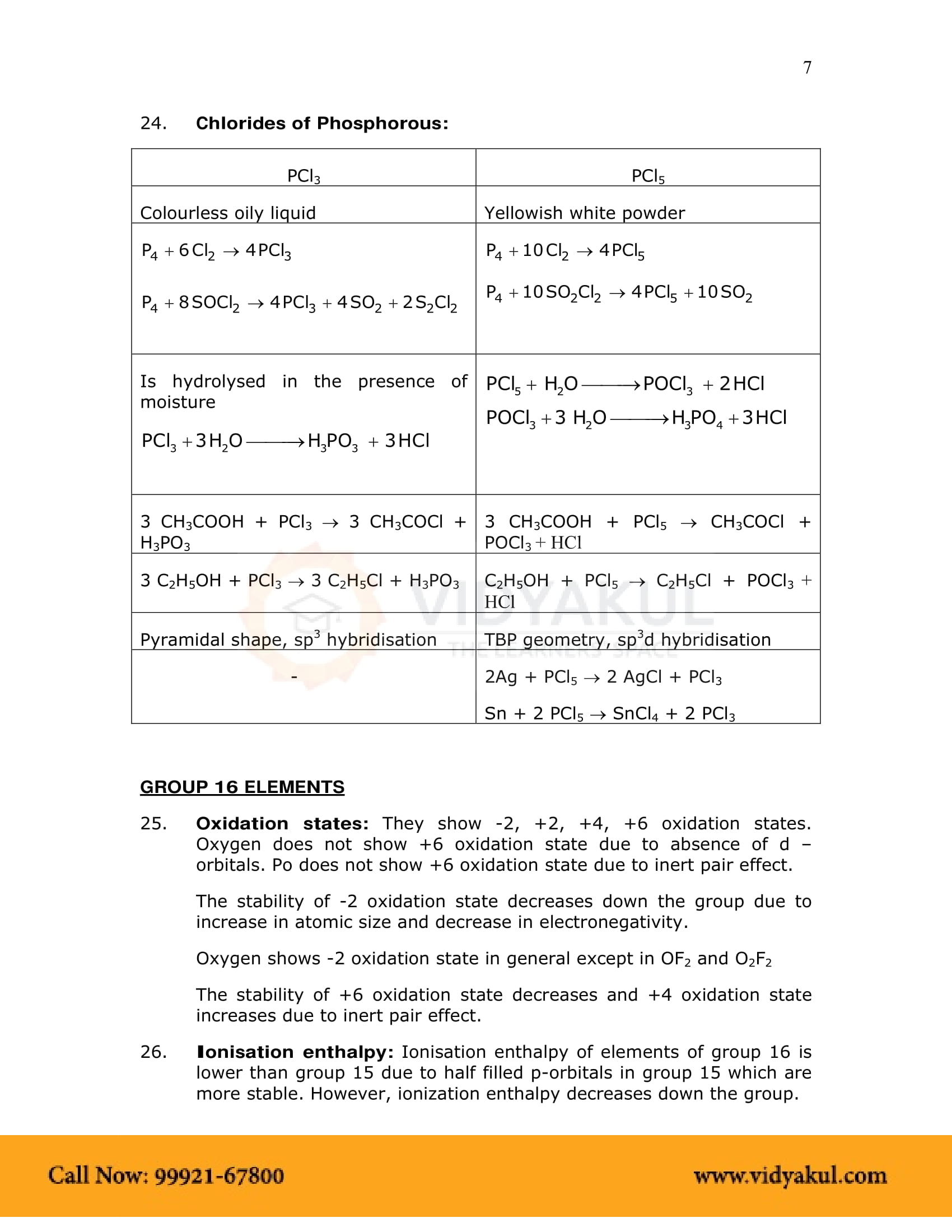
Does the reactivity of phosphorous differs from nitrogen? Explain in detail.
What are the different chemical reactions involved in the preparation of nitrogen in the laboratory?



